|
Here's something: If you have a concept of the size of an atom, and know that the atomic nucleus is about 10,000 times smaller with a volume a one-trillionth that of an atom then you should appreciate how much force it would take to push two uranium nuclei right up against each other. It's slightly less than 2,000 lbs! I happened to calculate this because the question came to mind while I was watching a show about the Large Hadron Collider. I had estimated a millionth of a pound, maybe even on the order of a thousandth of a pound, but, boy, was I surprised. I'm still not convinced my calculation is correct, even after posting it on Physics Forum and asking several physics-trained people about it and getting no objections. Here are some specifics on how I did the calculation: I Googled up "nuclear diameter" and got 15 femtometers (15x10-15 meter) from Wikipedia. Here's a diagram of two uranium nuclei pushed right up against each other: 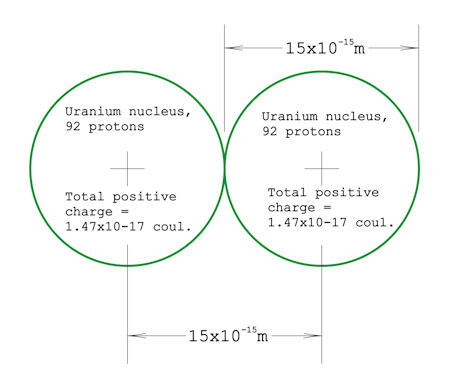
|